The Big Picture: Evolution of Extreme Structures
We study the development and evolution of extreme morphologies in insects, such as the elaborate horns and mandibles of beetles. The chafers, or scarabs, are notorious for the diversity and extravagance of their weapons. Within the superfamily three lineages appear to have independently evolved extreme fighting structures in males: the stag beetles (Lucanidae), the dung beetles (Scarabaeinae), and the rhinoceros beetles (Dynastinae), permitting us to compare how and why exaggerated male weapons evolved in each instance.

How Do You Build a Beetle Horn?
For more than 15 years our lab, our Co-PI (Laura Corley-Lavine, Washington State University), our colleagues and collaborators in Japan (Teruyuki Niimi, Hiroki Gotoh, and Shuji Shigenobu, National Institute for Basic Biology, Okazaki), and other labs (Armin Moczek, Indiana University), have studied the development of these structures, and a coherent and comprehensive picture has now emerged. In all three lineages the exaggerated weapons are sexually dimorphic, and sex-specific expression is achieved through the coupling of trait growth with the doublesex (dsx) pathway. Dsx regulates sex-specific development of genitalia and sexually selected structures (e.g., male sex combs) and behaviors (e.g., male courtship) in Drosophila, as well as a range of insect taxa. Males and females express alternate splice forms of dsx, and these act as transcription factors initiating sex-specific cascades of downstream gene expression.
In stag beetles, dsx modulates the amount of mandible growth by increasing (males) or decreasing (females) cellular sensitivity to a nutrition- and condition-sensitive hormone, JH. In both the dung and rhinoceros beetles the exaggerated weapons arose de novo through the co-option of upstream portions of the appendage patterning pathway, interacting with sex-specific splice forms of dsx, to initiate and coordinate a proximal-to-distal axis of outgrowth on the head and/or thorax. We suspect that ancestral scarabs already had the developmental capacity to uncouple growth of structures (e.g., genitalia) in males and females via the dsx pathway, and in both the dung and rhinoceros beetles mutations arose that coupled male-specific dsx splice forms with the appendage patterning pathway to generate novel weapons with sex-specific expression.
Our phylogenetic comparative studies of dung beetle horn evolution show that once present, horns were tremendously evolutionarily malleable, shifting in shape, relative size, and even location on the body, resulting in a rich diversity of extreme weapon forms. Given the ancient nature of these appendage patterning pathways and the conserved function of many of the involved genes, we are at last in a position to make a priori predictions for how these changes in horn form arise and, given the feasibility of multi-gene knockdowns and the looming potential of CRISPR/Cas9 (being developed for Trypoxylus by our colleagues in Japan), these ideas should be testable in the future.
Collaborators:
Laura Corley-Lavine (Washington State University)
Hiroki Gotoh (Hokkaido University)
Teruyuki Niimi & Shuji Shigenobu (National Institute for Basic Biology, Okazaki)
For more than 15 years our lab, our Co-PI (Laura Corley-Lavine, Washington State University), our colleagues and collaborators in Japan (Teruyuki Niimi, Hiroki Gotoh, and Shuji Shigenobu, National Institute for Basic Biology, Okazaki), and other labs (Armin Moczek, Indiana University), have studied the development of these structures, and a coherent and comprehensive picture has now emerged. In all three lineages the exaggerated weapons are sexually dimorphic, and sex-specific expression is achieved through the coupling of trait growth with the doublesex (dsx) pathway. Dsx regulates sex-specific development of genitalia and sexually selected structures (e.g., male sex combs) and behaviors (e.g., male courtship) in Drosophila, as well as a range of insect taxa. Males and females express alternate splice forms of dsx, and these act as transcription factors initiating sex-specific cascades of downstream gene expression.
In stag beetles, dsx modulates the amount of mandible growth by increasing (males) or decreasing (females) cellular sensitivity to a nutrition- and condition-sensitive hormone, JH. In both the dung and rhinoceros beetles the exaggerated weapons arose de novo through the co-option of upstream portions of the appendage patterning pathway, interacting with sex-specific splice forms of dsx, to initiate and coordinate a proximal-to-distal axis of outgrowth on the head and/or thorax. We suspect that ancestral scarabs already had the developmental capacity to uncouple growth of structures (e.g., genitalia) in males and females via the dsx pathway, and in both the dung and rhinoceros beetles mutations arose that coupled male-specific dsx splice forms with the appendage patterning pathway to generate novel weapons with sex-specific expression.
Our phylogenetic comparative studies of dung beetle horn evolution show that once present, horns were tremendously evolutionarily malleable, shifting in shape, relative size, and even location on the body, resulting in a rich diversity of extreme weapon forms. Given the ancient nature of these appendage patterning pathways and the conserved function of many of the involved genes, we are at last in a position to make a priori predictions for how these changes in horn form arise and, given the feasibility of multi-gene knockdowns and the looming potential of CRISPR/Cas9 (being developed for Trypoxylus by our colleagues in Japan), these ideas should be testable in the future.
Collaborators:
Laura Corley-Lavine (Washington State University)
Hiroki Gotoh (Hokkaido University)
Teruyuki Niimi & Shuji Shigenobu (National Institute for Basic Biology, Okazaki)

What Makes a Horn (Or Any Ornament or Weapon) an Honest Signal?
The most elaborate ornaments and weapons of sexual selection often function as signals, accurately revealing the resource holding potential of a rival male, or the size, status, and physiological condition of a potential mate. Decades of theoretical and empirical studies now show that despite an incredible breadth of materials (skin, bone, feather, chitin) and body parts (horns, tails, fins, beaks, legs, dewlaps) all of these signaling structures share three essential properties: they are larger/ more conspicuous than other surrounding structures; they are more variable in their expression from male to male than are other structures; and their growth is more sensitive to nutrition, stress, parasites, and body condition than is the growth of other body structures. Combined, these three properties (exaggerated size, hypervariability, and heightened condition-sensitive expression) make these particular traits more informative to receivers than other nearby body parts, favoring males or choosy females who pay attention to them.
We used the Japanese rhinoceros beetle Trypoxylus dichotomus as a pilot system to begin to explore, for the first time in any animal, the developmental and genetic regulatory mechanisms responsible for these critical properties of ornament/weapon expression. Using other structures in the same animals (wings, genitalia) as controls, we searched for unique patterns of gene expression associated with exaggerated growth, hypervariability, and heightened condition-sensitivity. Horns, wings, and genitalia grow at the same time (a burst of cell division at the end of the larval period), and all are exposed as they grow to the same circulating milieu of nutrients and hormones. Yet the horns respond to this hemolymph environment differently from the other traits - they are more sensitive to subtle differences in the nutritional state, size, and health of the animal than are the other structures. Using whole-transcriptome comparisons of gene expression in horns, wings, and genitalia of high and low nutrition animals, and RNAi knockdowns of identified candidate genes, we showed that in Trypoxylus beetles the insulin/IGF pathway acts as a whole-animal signal of individual body condition, modulating the rate and amount of growth of adult tissues in a coordinated and body size-specific fashion. Horns, we found, differ from other structures because cells in the developing horns are more sensitive to variations in these circulating insulin/IGF signals than are cells in the other traits, resulting in rapid growth of horns in the largest, best-condition males (exaggerated trait size); suppressed growth of horns in the smallest males (i.e., a wider range among males in horn length, or hypervariability); and increased nutrition-sensitivity of horns relative to other structures (heightened condition-sensitive expression).
We now know that it isn't always the insulin/IGF pathway. Indeed, in stag beetle mandibles heightened condition-sensitive growth results from increases in cellular sensitivity to JH, a different endocrine signal of nutritional state. And in dung beetles horns appear to be extra sensitive to both insulin/IGF and JH. But the glimpse into mechanism provided by our work helped show how coordinated suites of pleiotropic characteristics, all predicted to evolve in the context of signaling and sexual selection, can arise through the same simple change in an underlying developmental mechanism. It is now clear that insulin/IGF signaling influences the expression of cervid antlers, enlarged male chelae in shrimp and crabs, avian plumage ornaments, and even birdsong and male insect cuticular hydrocarbon (CHC) profiles. Heightened conditional expression of all of these sexually selected signal traits appears to result from an increase in their sensitivity to circulating levels of insulin/IGF. Consequently, we now have a working hypothesis for how beetle horns arose, for how they subsequently became exaggerated, and for how horns, and indeed many other exaggerated ornaments and weapons of sexual selection, became honest signals of resource holding potential, status, health, and/or genetic quality.
The most elaborate ornaments and weapons of sexual selection often function as signals, accurately revealing the resource holding potential of a rival male, or the size, status, and physiological condition of a potential mate. Decades of theoretical and empirical studies now show that despite an incredible breadth of materials (skin, bone, feather, chitin) and body parts (horns, tails, fins, beaks, legs, dewlaps) all of these signaling structures share three essential properties: they are larger/ more conspicuous than other surrounding structures; they are more variable in their expression from male to male than are other structures; and their growth is more sensitive to nutrition, stress, parasites, and body condition than is the growth of other body structures. Combined, these three properties (exaggerated size, hypervariability, and heightened condition-sensitive expression) make these particular traits more informative to receivers than other nearby body parts, favoring males or choosy females who pay attention to them.
We used the Japanese rhinoceros beetle Trypoxylus dichotomus as a pilot system to begin to explore, for the first time in any animal, the developmental and genetic regulatory mechanisms responsible for these critical properties of ornament/weapon expression. Using other structures in the same animals (wings, genitalia) as controls, we searched for unique patterns of gene expression associated with exaggerated growth, hypervariability, and heightened condition-sensitivity. Horns, wings, and genitalia grow at the same time (a burst of cell division at the end of the larval period), and all are exposed as they grow to the same circulating milieu of nutrients and hormones. Yet the horns respond to this hemolymph environment differently from the other traits - they are more sensitive to subtle differences in the nutritional state, size, and health of the animal than are the other structures. Using whole-transcriptome comparisons of gene expression in horns, wings, and genitalia of high and low nutrition animals, and RNAi knockdowns of identified candidate genes, we showed that in Trypoxylus beetles the insulin/IGF pathway acts as a whole-animal signal of individual body condition, modulating the rate and amount of growth of adult tissues in a coordinated and body size-specific fashion. Horns, we found, differ from other structures because cells in the developing horns are more sensitive to variations in these circulating insulin/IGF signals than are cells in the other traits, resulting in rapid growth of horns in the largest, best-condition males (exaggerated trait size); suppressed growth of horns in the smallest males (i.e., a wider range among males in horn length, or hypervariability); and increased nutrition-sensitivity of horns relative to other structures (heightened condition-sensitive expression).
We now know that it isn't always the insulin/IGF pathway. Indeed, in stag beetle mandibles heightened condition-sensitive growth results from increases in cellular sensitivity to JH, a different endocrine signal of nutritional state. And in dung beetles horns appear to be extra sensitive to both insulin/IGF and JH. But the glimpse into mechanism provided by our work helped show how coordinated suites of pleiotropic characteristics, all predicted to evolve in the context of signaling and sexual selection, can arise through the same simple change in an underlying developmental mechanism. It is now clear that insulin/IGF signaling influences the expression of cervid antlers, enlarged male chelae in shrimp and crabs, avian plumage ornaments, and even birdsong and male insect cuticular hydrocarbon (CHC) profiles. Heightened conditional expression of all of these sexually selected signal traits appears to result from an increase in their sensitivity to circulating levels of insulin/IGF. Consequently, we now have a working hypothesis for how beetle horns arose, for how they subsequently became exaggerated, and for how horns, and indeed many other exaggerated ornaments and weapons of sexual selection, became honest signals of resource holding potential, status, health, and/or genetic quality.
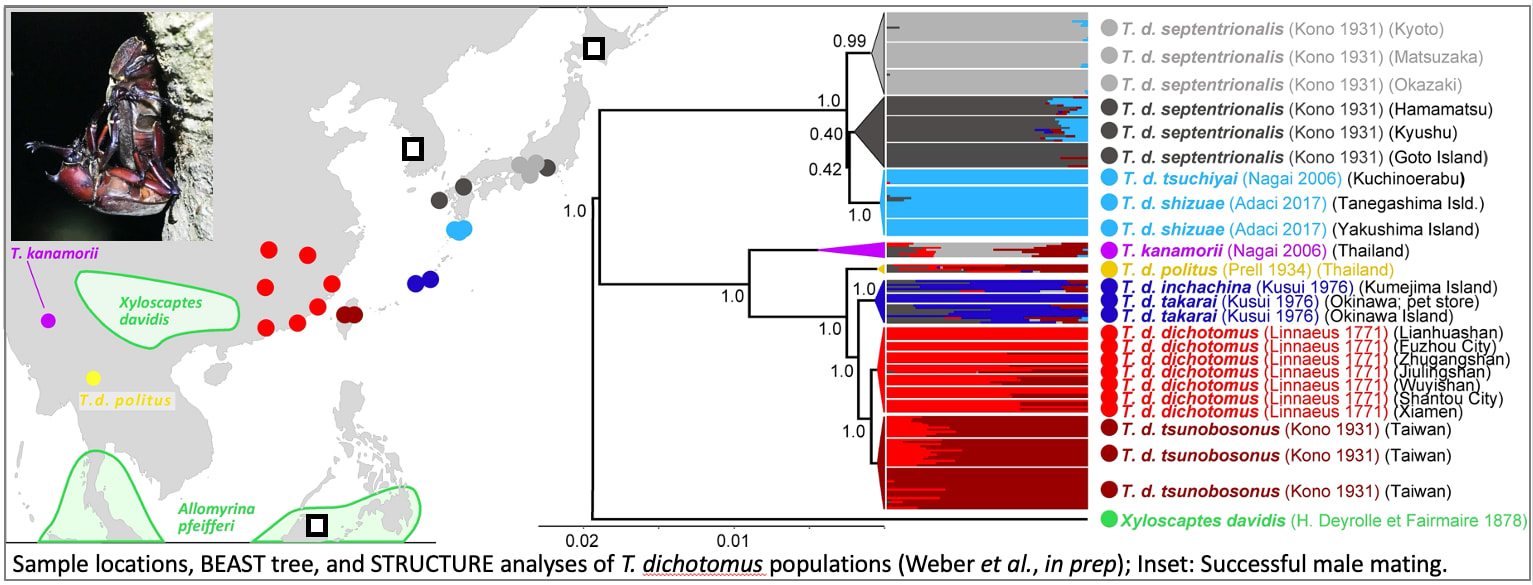
How Have Beetle Horns Evolved?
After years spent studying the mechanisms of horn development, we shifted our focus to begin to examine how and why these weapons evolve. Using genomic resources we and our collaborators developed for T. dichotomus, including a re-sequenced genome (Phase Genomics Hi-C, 10 chromosomes, 552.2 Mb) and 50,000 ddRADseq markers, we sampled 300 beetles from 22 locations across the range of this species. Our sample locations represented eight genetically distinct populations and, including DNA from the closest sister species, Xyloscaptes davidis, we were able to produce a well-supported hypothesis for the historical relationships among these populations.
We are using this tree to reconstruct changes in the relative length and mechanical lifting strength of male horns. Working with Co-PI Brook Swanson (Gonzaga University), we show that contemporary populations vary impressively and at least partially independently in horn length and strength. To begin to discern how these changes were brought about, we identified a pair of sister populations that differed dramatically in horn length (Honshu [long] and Yakushima [short]), sequenced the genomes of 70 Honshu and 50 Yakushima animals using PoolSeq, and scanned for FST outliers ('footprints' of strong recent selection). Approximately 50 genes fell within these outlier regions. We compared these with a priori predictions based on our prior evo-devo work, identified the highest priority candidates, and with our collaborators in Japan we will begin to knock these genes down using RNAi to test for functional links with horn growth. We also used RNAseq to contrast gene expression in developing horns from these same two long and short horned populations. Candidate genes identified from these analyses will be mapped to the genome and aligned with the "footprints of selection" analyses above.
Collaborators:
Brook Swanson (Gonzaga University)
Jesse Weber (University of Wisconsin)
Wataru Kojima (Yamaguchi University)
Kunio Araya (Kyushu University)
Erica Larson (University of Denver)
After years spent studying the mechanisms of horn development, we shifted our focus to begin to examine how and why these weapons evolve. Using genomic resources we and our collaborators developed for T. dichotomus, including a re-sequenced genome (Phase Genomics Hi-C, 10 chromosomes, 552.2 Mb) and 50,000 ddRADseq markers, we sampled 300 beetles from 22 locations across the range of this species. Our sample locations represented eight genetically distinct populations and, including DNA from the closest sister species, Xyloscaptes davidis, we were able to produce a well-supported hypothesis for the historical relationships among these populations.
We are using this tree to reconstruct changes in the relative length and mechanical lifting strength of male horns. Working with Co-PI Brook Swanson (Gonzaga University), we show that contemporary populations vary impressively and at least partially independently in horn length and strength. To begin to discern how these changes were brought about, we identified a pair of sister populations that differed dramatically in horn length (Honshu [long] and Yakushima [short]), sequenced the genomes of 70 Honshu and 50 Yakushima animals using PoolSeq, and scanned for FST outliers ('footprints' of strong recent selection). Approximately 50 genes fell within these outlier regions. We compared these with a priori predictions based on our prior evo-devo work, identified the highest priority candidates, and with our collaborators in Japan we will begin to knock these genes down using RNAi to test for functional links with horn growth. We also used RNAseq to contrast gene expression in developing horns from these same two long and short horned populations. Candidate genes identified from these analyses will be mapped to the genome and aligned with the "footprints of selection" analyses above.
Collaborators:
Brook Swanson (Gonzaga University)
Jesse Weber (University of Wisconsin)
Wataru Kojima (Yamaguchi University)
Kunio Araya (Kyushu University)
Erica Larson (University of Denver)

Why Have Beetle Horns Diverged in Length?
The Japanese rhinoceros beetle has been studied most extensively on Honshu Island, Japan, where adults emerge from the soil during summer months, fly to wounds on the sides of mature oak and maple trees, and feed on oozing sap. Males arrive at the feeding ground first and battle for possession of the feeding territories, releasing aggregation pheromones which, as in other scarabs, attract both females and rival males to the area. Breeding populations congregate around a few host trees, and these local populations are separated by many miles from other breeding aggregations, making it feasible to mark and follow all beetles in the population.
Fights between males are frequent, and males with relatively large body sizes and long horn lengths are most likely to win ownership of feeding territories. Females mate with males when they visit these territories to feed, and in Kyoto (Honshu Island), the first wild population where mating success was quantified, males with the longest horns mated with the most females (work by Yoshihito Hongo). Surprisingly, female T. dichotomus mate just once, with a single male, meaning that, to a remarkable degree it is possible to quantify male lifetime reproductive success (e.g., total selection on horns) in the wild simply by recording the total number of successful mating events over a breeding season.
In collaboration with Yoshihito Hongo, we tested whether local breeding conditions and population differences in the strength of sexual selection might help explain recent evolutionary shifts in relative horn length. Specifically, we conducted full breeding season studies of fighting and mating success at four additional populations, and combined our results with data from Hongo's original population (Kyoto, Japan). Included populations differ in their respective scaling relationships such that males in three populations (Puli & Chia-yi, Taiwan, and Yakushima Island, Japan) have relatively shorter horn lengths than males in the remaining two (Kameoka & Kyoto, Honshu Island, Japan). We found that sexual selection was strong and directional in both of the long horned populations, and relaxed or nonexistent at the short horned populations. Furthermore, our field studies point to a putative ecological driver of rapid, recent weapon divergence: geographic variation in the extent to which feeding territories are limiting. We will be conducting field studies at additional locations, adding to this emerging picture of ecology and sexual selection.
Collaborators:
Yoshihito Hongo (Kyoto University)
Wataru Kojima (Yamaguchi University)
Brook Swanson (Gonzaga University)
The Japanese rhinoceros beetle has been studied most extensively on Honshu Island, Japan, where adults emerge from the soil during summer months, fly to wounds on the sides of mature oak and maple trees, and feed on oozing sap. Males arrive at the feeding ground first and battle for possession of the feeding territories, releasing aggregation pheromones which, as in other scarabs, attract both females and rival males to the area. Breeding populations congregate around a few host trees, and these local populations are separated by many miles from other breeding aggregations, making it feasible to mark and follow all beetles in the population.
Fights between males are frequent, and males with relatively large body sizes and long horn lengths are most likely to win ownership of feeding territories. Females mate with males when they visit these territories to feed, and in Kyoto (Honshu Island), the first wild population where mating success was quantified, males with the longest horns mated with the most females (work by Yoshihito Hongo). Surprisingly, female T. dichotomus mate just once, with a single male, meaning that, to a remarkable degree it is possible to quantify male lifetime reproductive success (e.g., total selection on horns) in the wild simply by recording the total number of successful mating events over a breeding season.
In collaboration with Yoshihito Hongo, we tested whether local breeding conditions and population differences in the strength of sexual selection might help explain recent evolutionary shifts in relative horn length. Specifically, we conducted full breeding season studies of fighting and mating success at four additional populations, and combined our results with data from Hongo's original population (Kyoto, Japan). Included populations differ in their respective scaling relationships such that males in three populations (Puli & Chia-yi, Taiwan, and Yakushima Island, Japan) have relatively shorter horn lengths than males in the remaining two (Kameoka & Kyoto, Honshu Island, Japan). We found that sexual selection was strong and directional in both of the long horned populations, and relaxed or nonexistent at the short horned populations. Furthermore, our field studies point to a putative ecological driver of rapid, recent weapon divergence: geographic variation in the extent to which feeding territories are limiting. We will be conducting field studies at additional locations, adding to this emerging picture of ecology and sexual selection.
Collaborators:
Yoshihito Hongo (Kyoto University)
Wataru Kojima (Yamaguchi University)
Brook Swanson (Gonzaga University)
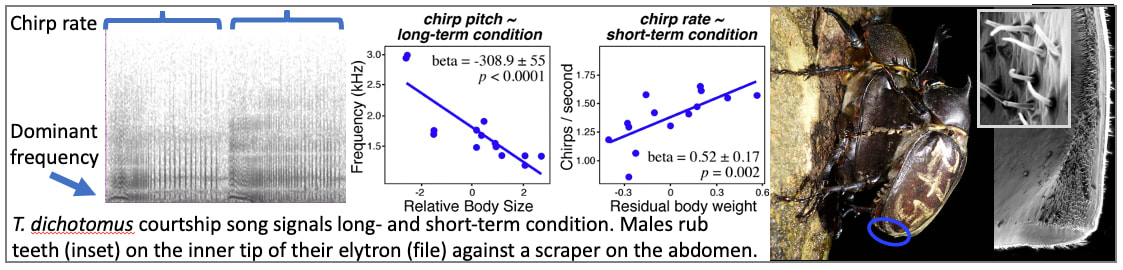
Why do beetles sing and dance?
While conducting focal animal observations in the field, we noticed a number of surprising behaviors. First, females often rejected the mating attempts of males, even if they were large, long-horned males and legitimate territory holders. Female rhinoceros beetles have to slide abdominal plates aside to permit aedeagal access, and therefore control copulation, and our crews repeatedly observed females denying access to males. Likely in response to this, territorial males spent many hours each night courting females, including engaging in repeated bouts of stridulatory singing. We now suspect females use a combination of courtship songs and trembling “dances” to determine whether or not to accept a mate, adding a surprising twist to this otherwise-textbook resource-defense mating system. Our current experiments explore why females might be rejecting the mating attempts of territory-holding males; how they base their mate-choice decisions; and, why they aren't using the size of the male horn as a reliable signal of male quality.
Collaborators:
Wataru Kojima (Yamaguchi University)
Leigh Simmons (University of Western Australia)
